 
These are part of a greater family of other chlorine oxides. The molecular weight of the compound is 83.45 g/mol. Chlorine trioxide is non-combustible in nature but when it gets combined with combustible materials explosion is likely to occur. Several oxyanions of chlorine exist, in which it can assume oxidation states of −1, +1, +3, +5, or +7 within the corresponding anions Cl −, ClO −, ClO −Ĥ, known commonly and respectively as chloride, hypochlorite, chlorite, chlorate, and perchlorate. Obviously the bonding in the chlorate ion is not ionic but this procedure allows you to construct 'half-equations' and then an overall equation. ClO3- or chlorine trioxide or chlorate is a monovalent inorganic anion that is soluble in water. The most important chlorite is sodium chlorite (NaClO 2) this is used in the bleaching of textiles, pulp, and paper, however despite its strongly oxidizing nature it is often not used directly being instead used to generate the neutral species chlorine dioxide (ClO 2), normally via a reaction with HCl:ĥ NaClO 2 + 4 HCl → 5 NaCl + 4 ClO 2 + 2 H 2O Other oxyanions Ĭhlorite is the strongest oxidiser of the chlorine oxyanions on the basis of standard half cell potentials. The chlorite ion adopts a bent molecular geometry, due to the effects of the lone pairs on the chlorine atom, with an O–Cl–O bond angle of 111° and Cl–O bond lengths of 156 pm. First, the explosively unstable gas chlorine dioxide, ClO 2 is produced by reducing sodium chlorate with a suitable reducing agent such as methanol, hydrogen peroxide, hydrochloric acid or sulfur dioxide. 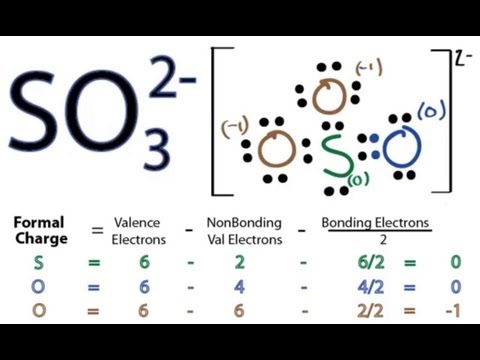
Sodium chlorite is derived indirectly from sodium chlorate, NaClO 3. Heavy metal chlorites (Ag +, Hg +, Tl +, Pb 2+, and also Cu 2+ and NH +Ĥ) are unstable and decompose explosively with heat or shock. The alkali metal and alkaline earth metal compounds are all colorless or pale yellow, with sodium chlorite (NaClO 2) being the only commercially important chlorite. Since it cannot be concentrated, it is not a commercial product. while hypochlorite ion is linear and perchlorate ion is tetrahedral and there is no effect of lone pair on them. The industrial scale synthesis for sodium chlorate starts from sodium chloride.The free acid, chlorous acid HClO 2, is the least stable oxoacid of chlorine and has only been observed as an aqueous solution at low concentrations. in chlorate ion bond angle changes due to presence of lone pair on chlorine atom.  
Metal chlorates can be prepared by adding chlorine to hot metal hydroxides, for example, KClO 3: 3Cl 2 + 6KOH → 5KCl + KClO 3 + 3H 2O Most pyrotechnic applications which used chlorates in the past now use perchlorates instead. Chlorates were once widely used in pyrotechnics, though their use has fallen due to their instability. As predicted by VSEPR, chlorate anions have trigonal pyrimidal structures.Ĭhlorates are powerful oxidizers and should be kept away from organics or easily oxidized materials. "Chlorate" can also refer to chemical compounds containing this anion chlorates are the salts of chloric acid. (3) The structure of perchloric acid is O Cl +7 Chlorate ion : 3 (ClO- ) O Cl O O Trigonal pyramidal O OH. In this case, the chlorine atom is in the +5 oxidation state. (a) Due to highest bond dissociation energy. The chlorate anion has the formula ClO 3 −. The global Sodium Chlorate market size was valued at USD million in 2022 and is forecast to a readjusted size of USD million by 2030 with a CAGR during analysis period. Structure and bonding in the chlorate ion 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
